Into The WIld
Final Documentary
For this project we did a lot. On the humanities side we started off by reading John Krakauer's Into The Wild. We read the chapters one by one and answered questions about them in our composition books. In Physics we were taught about tents, coolers, headlamps and soda can stoves. We wrote diagrams, storys, how-to's and many other things all in out copmosition books. In January we went on a two day trip to Anza-Borrego to experience the wild. Eventually we put all of the contents of the composition book into a final journal. My special role in this project was to document it. Me and four other people were chosen to film and put together a documentary. I was in charge of writing the narration for it. All in all the project was lots of fun and I learned a lot from the camping trip.
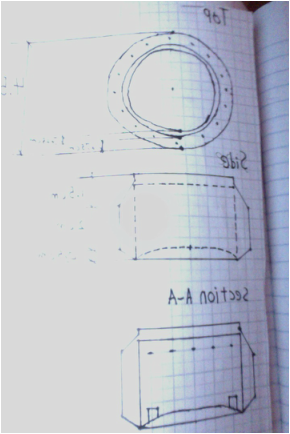
We also needed to write a soda can spec sheet which I put below...
We also needed to write a soda can spec sheet which I put below...
Tech Piece
How it's made
To build my soda can stove I used a box cutter, pliers, scissors, ruler, sharpie and a pushpin. First I used the box cutter that was taped to a block of wood to cut four centimeters off the bottom half of the can. Next I flipped the can over and cut the top four centimeters off. I cut the middle piece down the middle and around a four centimeters width. When I had the three sections of the can separated I carved the top of the can. So the top part was a cylinder with no bottom or top. After that, I took my middle piece and cut two slits into each end. When you attach both slits, you make a smaller circle. I cut three squares into the bottom of the strip to let the fuel flow between the inner and outer chamber. Once that was done, I poked sixteen holes on the upper sides of the top part of the can. I then put the strip in the top part of the can. I proceeded to crimp the bottom part’s edges so it can fit into the top part. I put them into each other and taped them together. That’s how I built my stove! I think the over all building process was solid. I had no problem making it and I had fun doing it.
How it works
A soda can stove’s inter-workings are compromised of an inner and outer chambers connected by three holes in the inter chamber’s walls. To work, you need to put liquid fuel into the both the chambers. When you have fuel you light the inner chamber on fire. As the liquid fuel heats up in the inner chamber, the vapor pressure of the fuel increases making it boil. When the vapor pressure increases the walls of the inner chamber heat up causing the fuel in the outer chamber to heat up. When it boils pressure builds up in the outer chamber. As the pressure reaches a limit, jets of pressurized fuel shoot out from the holes in the outer chamber. The jets ignite from the center flame making a real stove. Heat from both sources of flames continues to heat the stove. This continues the cycle until the fuel is exhausted. Vaporized fuel will come out of the center chamber until you put a pot or other flat object on top of the opening. Vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid or solid. The pressure of the vapor is consequential from the evaporation of a liquid.
Methods of conduction
The water in a pot boils when the flame from the stovetop heats the pot, and the heat from the pot is transferred to the water via conduction. Conduction is the transfer of heat between two objects of a stationary system caused by a temperature difference between the objects. The molecules on an object move, it creates heat on an object, this builds kinetic energy. When the warm object touches another one of a lower temperature, the molecules from the first object transfer their kinetic energy to the colder object. This then raises the temperature of the object equal to the heat of the original object. So the pot gets hot, heating up the water inside. As the water heats, convection occurs. Convection is when the liquid expands and rises because the density lessens as it heats. As it rises, the cold denser water sinks towards bottom. Once that warms it rises and starts the cycle over again. Convection is the primary way heat moves through liquids and gasses.
How it's made
To build my soda can stove I used a box cutter, pliers, scissors, ruler, sharpie and a pushpin. First I used the box cutter that was taped to a block of wood to cut four centimeters off the bottom half of the can. Next I flipped the can over and cut the top four centimeters off. I cut the middle piece down the middle and around a four centimeters width. When I had the three sections of the can separated I carved the top of the can. So the top part was a cylinder with no bottom or top. After that, I took my middle piece and cut two slits into each end. When you attach both slits, you make a smaller circle. I cut three squares into the bottom of the strip to let the fuel flow between the inner and outer chamber. Once that was done, I poked sixteen holes on the upper sides of the top part of the can. I then put the strip in the top part of the can. I proceeded to crimp the bottom part’s edges so it can fit into the top part. I put them into each other and taped them together. That’s how I built my stove! I think the over all building process was solid. I had no problem making it and I had fun doing it.
How it works
A soda can stove’s inter-workings are compromised of an inner and outer chambers connected by three holes in the inter chamber’s walls. To work, you need to put liquid fuel into the both the chambers. When you have fuel you light the inner chamber on fire. As the liquid fuel heats up in the inner chamber, the vapor pressure of the fuel increases making it boil. When the vapor pressure increases the walls of the inner chamber heat up causing the fuel in the outer chamber to heat up. When it boils pressure builds up in the outer chamber. As the pressure reaches a limit, jets of pressurized fuel shoot out from the holes in the outer chamber. The jets ignite from the center flame making a real stove. Heat from both sources of flames continues to heat the stove. This continues the cycle until the fuel is exhausted. Vaporized fuel will come out of the center chamber until you put a pot or other flat object on top of the opening. Vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid or solid. The pressure of the vapor is consequential from the evaporation of a liquid.
Methods of conduction
The water in a pot boils when the flame from the stovetop heats the pot, and the heat from the pot is transferred to the water via conduction. Conduction is the transfer of heat between two objects of a stationary system caused by a temperature difference between the objects. The molecules on an object move, it creates heat on an object, this builds kinetic energy. When the warm object touches another one of a lower temperature, the molecules from the first object transfer their kinetic energy to the colder object. This then raises the temperature of the object equal to the heat of the original object. So the pot gets hot, heating up the water inside. As the water heats, convection occurs. Convection is when the liquid expands and rises because the density lessens as it heats. As it rises, the cold denser water sinks towards bottom. Once that warms it rises and starts the cycle over again. Convection is the primary way heat moves through liquids and gasses.
Reflection
I thought I did well on this project. My job was to document the project. I had a lot of fun and I think that reflected on my efforts. I worked well with my group and finished requirments on time. I loved this project and I hope to do something like it again.
My soda can stove drawings